Depending on the member’s plan, we may require prior authorization or prior approval for genetic testing.
| For these members | Authorization (or prior approval) is required | Who to contact for prior authorization (or prior approval) |
|---|---|---|
|
Yes (for most tests – see below) | Carelon Medical Benefits Management |
|
Yes (some tests) | Blue Cross Blue Shield of Massachusetts Clinical Intake department at 1-800-689-7219 |
Prior authorization program with Carelon Medical Benefits Management
For members requiring prior authorization with Carelon (see the table above), here’s some information about the program.
As always, we recommend checking benefits and eligibility to determine the member’s benefits and any authorization requirements before performing services.
Refer to Carelon Genetic Testing Management Program CPT and HCPCS Codes medical policy 957.
Categories of tests that require prior authorization include, but may not be limited to:
- Genetic testing for hereditary risk of cardiac disease
- Genetic testing for hereditary cancer susceptibility
- Genetic testing for single-gene and multifactorial conditions
- Pharmacogenetic and thrombophilia genetic testing
- Reproductive carrier screening and prenatal diagnosis
- Solid and hematologic tumor and malignancy testing
- Whole exome and genome sequencing
Prior authorization is not required for genetic testing associated with organ transplantation.
For preimplantation genetic testing, we don’t require prior authorization with Carelon; however, you should continue to request prior authorization from Blue Cross Blue Shield of Massachusetts for biopsy of the embryo. Refer to our medical policy, Preimplantation Genetic Testing: 088.
Ordering physicians or clinicians must request authorization before the member receives the test (or before performing services). There are three ways to contact Carelon to request prior authorization:
- Through a direct link on this site (no additional registration is required). In the eTools tab, go to Carelon. Click the Go Now button.
- Go directly to the Carelon ProviderPortal (registration is required)
- Go to www.providerportal.com
- Call Carelon’s Contact Center at 1-866-745-1783
- Available Monday through Friday, 8 a.m. – 6 p.m. If you leave a voicemail after these hours, Carelon will respond the next business day.
Here’s some general information you may need to request the authorization. You can find checklists for specific tests on the Carelon genetic testing program site.
- Member’s first and last name, date of birth
- Summary of the patient’s clinical diagnosis
- Ordering provider’s first and last name
- Clinical summary from the genetic counseling appointment
- Test being requested and the name of the laboratory performing the test (you’ll be able to choose the test from a menu that includes branded panels, or you can manually enter it)
- Pedigree or summary of three-generation maternal and paternal family history
- Maternal and paternal ethnic background/race
- Date of service. This is the sample or collection date. For archival samples, the sample collection or retrieval date will serve as the date of service for review.
Once the ordering physician, clinician, or their office staff has entered the required information into the online authorization tool, you’ll get an immediate decision (in most cases). If Carelon needs more time to review the information, the system will indicate that it’s pending review. And, if Carelon needs more information for their review, you’ll get a request to submit additional information.
Please note: When you request prior authorization, it's for a specific genetic test. You aren't requesting prior authorization for the CPT code(s).
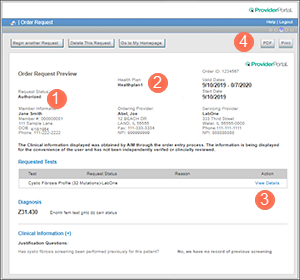
When your authorization is approved, Carelon ProviderPortal will indicate it’s "Authorized" in the Order Request Preview (see ![]() in the example below) You’ll also find:
in the example below) You’ll also find:
- The name of the approved test & CPT code(s)
- The units approved
Please be sure to:
- Tell the member that they must have the test within the timeframe approved (see
 in example). Otherwise, if the test is outside of those dates, a new authorization request will be needed.
in example). Otherwise, if the test is outside of those dates, a new authorization request will be needed.
- Attach a copy of the approved authorization to your lab requisition form. This will help the lab understand what's been approved. You’ll be able to download a PDF or print a copy (see
 in example) after you have submitted your authorization request and it’s been approved.
in example) after you have submitted your authorization request and it’s been approved.
- Click "View Details" (
 in example) to see all CPT codes associated with the test that are approved. Remember: You will need to make sure you have approval for each CPT code.
in example) to see all CPT codes associated with the test that are approved. Remember: You will need to make sure you have approval for each CPT code.
- Click "View Details" (
Important: We recommend attaching a copy of the approved authorization to your lab requisition instead of using a pre-populated lab requisition form, such as the one in the example below. When you enter the test into Carelon’s ProviderPortal, you are requesting the individual test, not a panel, so the test authorized and the pre-populated form are not always a match.

Please note: When you request prior authorization, it's for a specific genetic test. You aren't requesting prior authorization for the CPT code(s).
When a specimen comes into your lab for genetic testing, before you analyze the specimen, please remember to verify:
- An approved authorization for the test. You can verify the authorization approval by going to the Carelon ProviderPortal.
- The CPT codes approved as part of that authorization. Next, you can give those codes to your billing department for your claim, so your claims will process smoothly.
- Click "View Details" (See
 in the example above) to see any CPT codes associated with the test that are approved.
in the example above) to see any CPT codes associated with the test that are approved.
- Click "View Details" (See
We’re matching the approved CPT codes and unit amounts to the claim that the laboratory submits. If these fields don’t match, the claim will deny. To avoid a denied claim, we urge clinical and hospital laboratories to use the Carelon ProviderPortal to verify that an authorization is in place before the test is performed. We suggest sharing the authorization approval information (CPT codes and units) with your billing department.
We use Carelon clinical criteria to make medical necessity determinations. Refer to our medical policy 954 for links to Carelon’s criteria.
Blue Cross continues to maintain medical policies for the following:
- Preimplantation Genetic Testing: 088, for which the biopsy of the embryo will continue to require prior authorization directly from Blue Cross Blue Shield of Massachusetts, but the associated genetic testing does not require prior authorization if performed for preimplantation genetic diagnosis (as opposed to screening).
Carelon offers information on its resource website. You’ll find:
- Program information & clinical guidelines
- Tutorials (on how to enter an authorization using the Carelon online tool)
- Worksheets to help your office prepare the information needed to enter the authorization request